Pathways to electrolysis success for USA, Australia
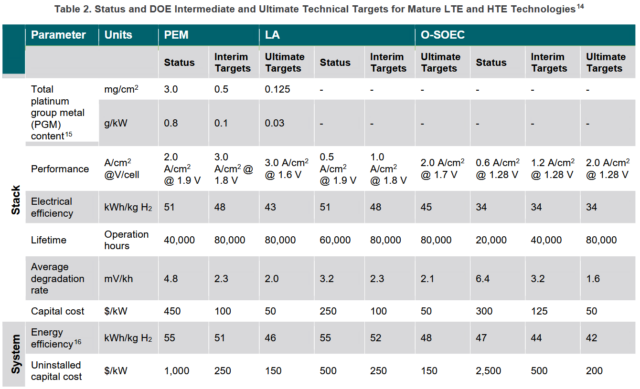
In order to achieve the aggressive Hydrogen Shot initiative goal of $1/kg production cost by 2030, the US Department of Energy sets out a series of performance and capital cost targets for established and lower-TRL electrolyser technologies. Meanwhile, Australia's national science agency CSIRO projects that Australia’s hydrogen electrolyser manufacturing sector could generate AU$1.7 billion in revenue annually by 2050.