Presentation
Sustainable Ammonia Production from Sun, Air and Water
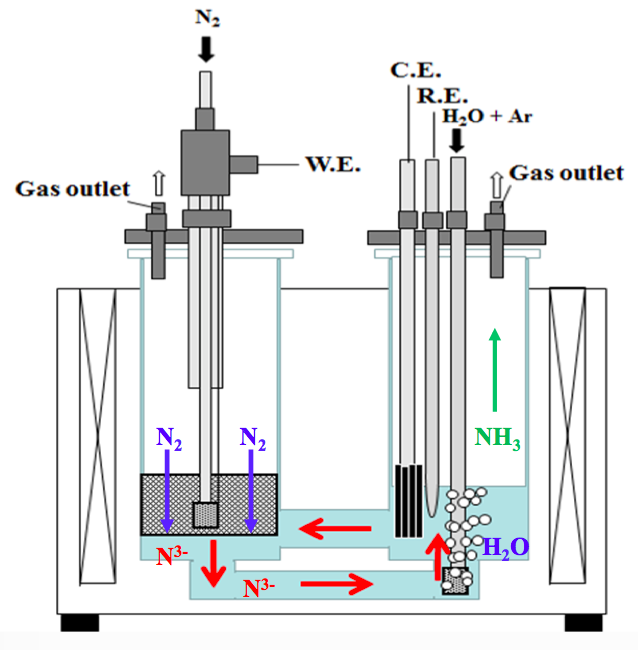
There is an ever growing demand for ammonia production that already reached globally 200 million tons per year by 2018 and is forecasted to increase to over 350 million tons per year by 2050 [1]. The application segment is dominated by the fertilizer industry, since the most important fertilizer and the world’s most widely produced chemical is urea. Ammonia is synthesized via the Haber-Bosch process, for which the required hydrogen and nitrogen are currently provided by using fossil fuels. This work proposes a novel approach to produce ammonia from the raw materials water and air only by utilizing solar energy…