Catalysts
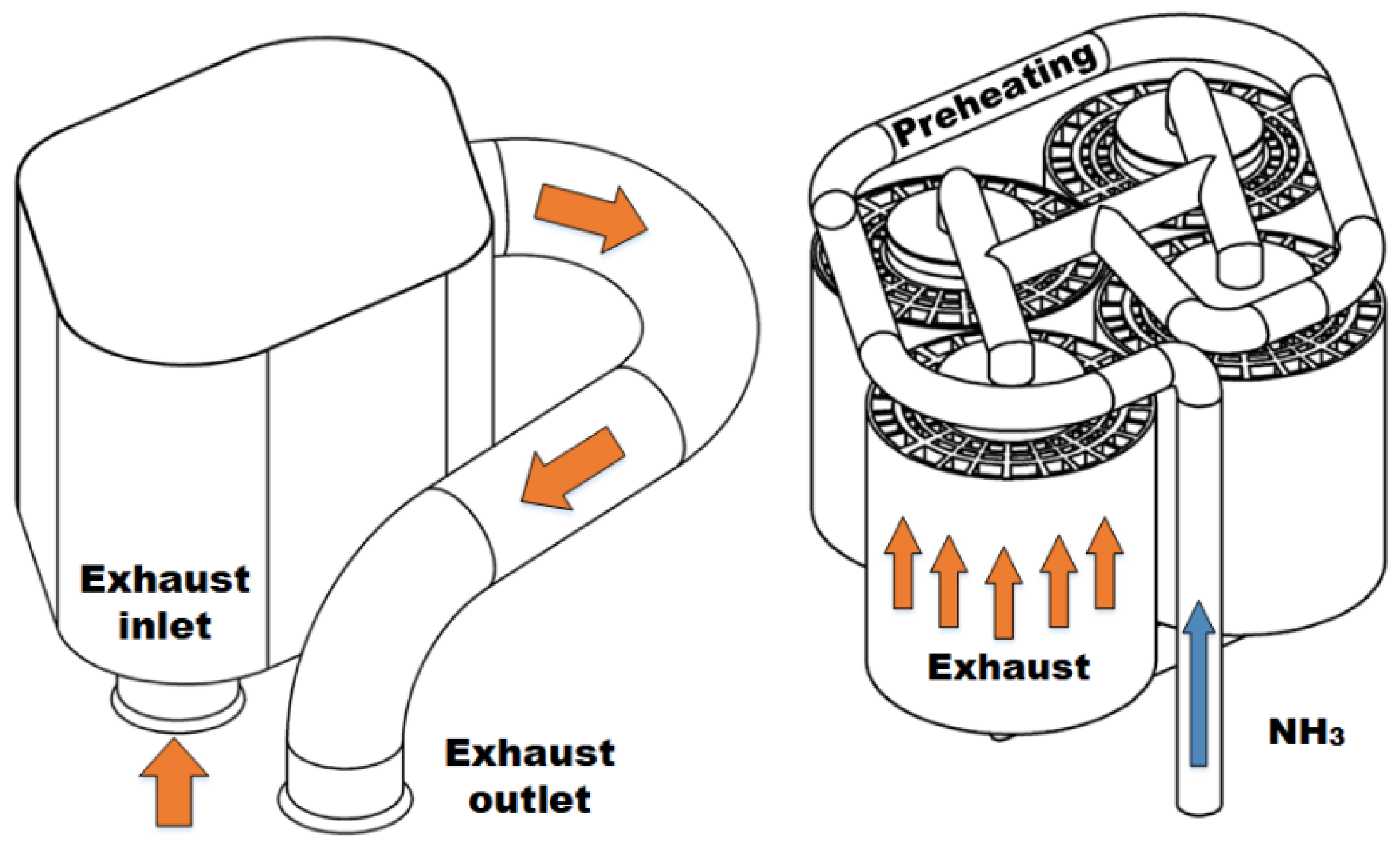
Exhaust gas treatment catalysts for ammonia-fueled engines
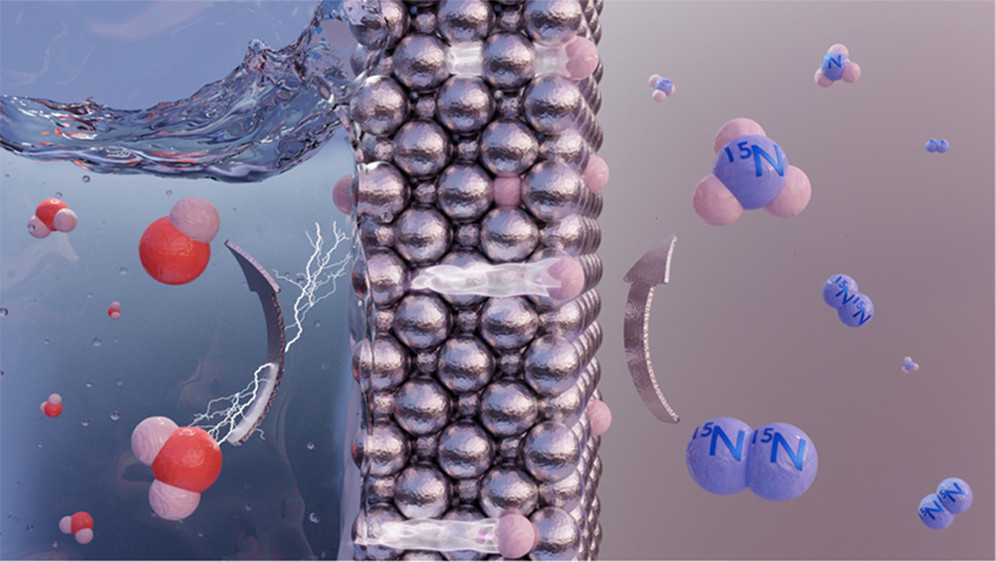
Advanced technologies powering green ammonia R&D
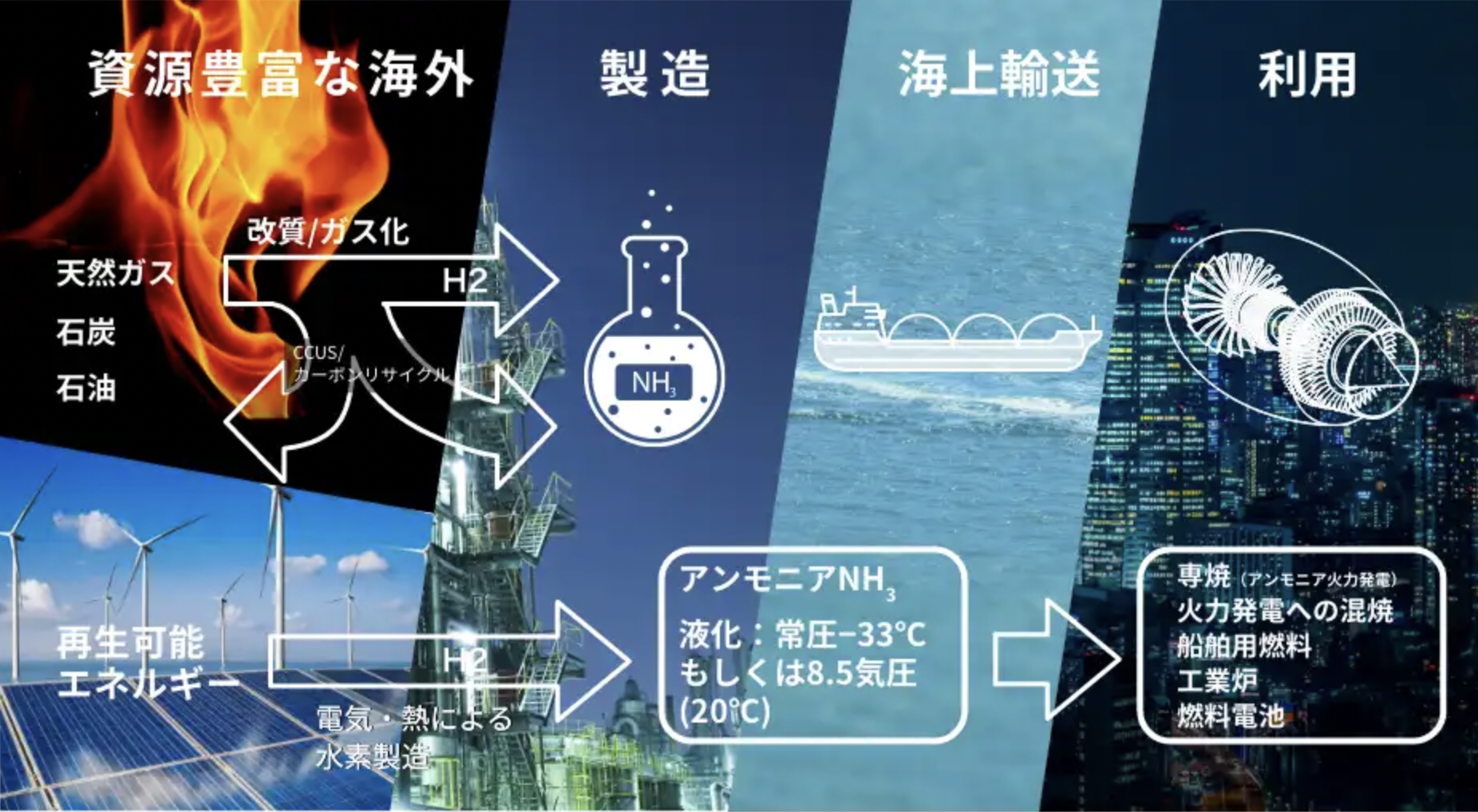
Sunborne Systems: bringing keystone technologies to market for the global zero-carbon energy infrastructure
AI & High-Performance Computing for ammonia catalyst R&D
Fujitsu and Iceland-based Atmonia will join forces to accelerate catalyst development for the production of ammonia via electrochemical nitrogen reduction reaction (eNRR). By using artificial intelligence and high-performance computing (HPC) technologies, the researchers can conduct “high-speed quantum chemical calculations” virtually rather than via physical experiments, allowing for greater flexibility & speed.
JERA targets 50% ammonia-coal co-firing by 2030
Japanese government funding via NEDO will support four critical ammonia energy projects, including JERA's new plan to demonstrate 50% ammonia-coal co-firing by 2030. Other projects include improved catalysts for ammonia production, low-temperature and low-pressure synthesis pathways, and developing 100% ammonia-fed boilers and gas turbines. In addition, a new cooperation agreement between ASEAN countries will see Japan support other members to adopt their ammonia energy solutions, particularly coal co-firing.
Ammonia combustion analysis: powertrains, turbines & power generation
This week we explore four updates in ammonia combustion R&D:
1. A team from the University of Cambridge has shown merchant vessels are the strongest candidates for conversion to run on ammonia powertrains, with cargo capacity losses of 4-9% able to be feasibly offset by operators.
2. Researchers at the University of Minnesota have successfully tested a thermochemical recuperation (TCR) reactor to improve the efficiency of a dual-fuel, diesel-ammonia compression ignition engine by minimising ammonia slip.
3. A global team led by Cardiff University researchers has revealed some of the inner workings of ammonia combustion in gas turbine flames.
4. A global team has produced a cradle-to-gate environmental assessment for ammonia production and ammonia-based electricity generation, suggesting that renewable and nuclear ammonia have a significant role to play in decarbonising the power sector.
3rd generation ammonia synthesis: new catalysts & production pathways
We look at four new developments this week:
1. A team from DTU Energy and the Dalian Institute of Chemical Physics have uncovered a new class of alternative catalysts for mild condition ammonia synthesis. The ternary ruthenium complex hydrides Li4RuH6 and Ba2RuH6 avoid the energy-intensive pathway of nitrogen dissociation in a "synergistic" manner.
2. A team from the Korea Institute of Machinery and Materials reported a highly selective (95%) plasma ammonia synthesis method.
3. A team from Delft University of Technology has presented an present an "unconventional electrochemical design" that physically separates hydrogen and dinitrogen activation sites.
4. A team at the Max Planck Institute for Coal Research has demonstrated a new mechanochemical ammonia synthesis system that operates at room temperature and pressures as low as 1 bar.
New UK joint venture for lightweight, modular ammonia crackers
Reaction Engines, IP Group, and the Science and Technology Facilities Council (STFC) launched a new joint venture this week at COP26 in Glasgow. The group will design and commercialise lightweight, modular ammonia cracking reactors to enable the use of ammonia in hard-to-decarbonise sectors, particularly aviation, shipping and off-grid power generation applications. The design will feature Reaction Engines’ heat exchanger technology developed for its SABRE™ air-breathing rocket engine. In this setup, exhaust heat is utilised to partially crack ammonia back into a fuel blend that "mimics" jet fuel. STFC will lead development of the cracking catalyst, with funding to be provided by IP Group.